Antibodies Revealing the Virus’s Weak Point: A Novel Neutralization Mechanism Against SARS‑CoV‑2
A new study published in PNAS identifies an unexpected mechanism that may reshape current understanding of how the immune system combats SARS‑CoV‑2—and potentially future viruses as well. The study was led by Akanksha Harit, a PhD candidate under the supervision of Prof. Moshe Dessau, head of the Laboratory for Structural Biology of Infectious Diseases, in collaboration with the laboratories of Prof. Meital Gal‑Tanamy and Prof. Natalie Freund at Tel Aviv University—an interdisciplinary effort that brings a novel perspective to the field of structural immunology.
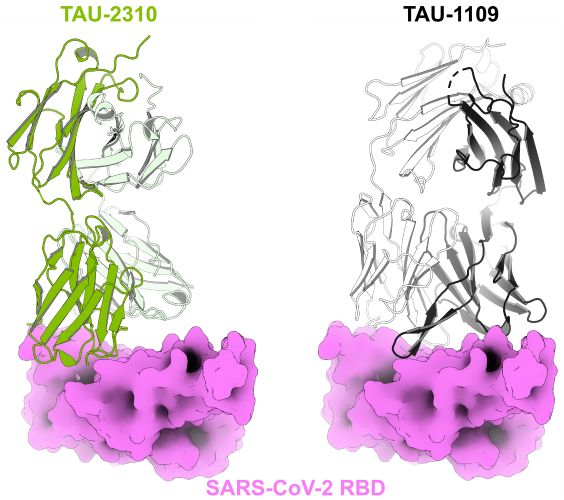
The researchers identified a novel neutralization mechanism mediated by monoclonal antibodies isolated from convalescent patients. These antibodies recognize “cryptic epitopes” on the viral spike protein—regions that are typically inaccessible to the immune system. Notably, these antibodies not only block viral function but also induce premature destabilization of the spike protein, which resides on the viral envelope and is responsible for host cell entry. By compromising the stability of the spike complex, the antibodies trigger early shedding of the S1 subunit, thereby abolishing the virus’s ability to enter host cells. This represents a particularly sophisticated mode of action, offering not only passive protection but active viral neutralization.
The implications extend well beyond SARS‑CoV‑2. The identified epitopes are evolutionarily conserved, meaning they remain largely unchanged across viral variants. This makes them highly promising targets for the development of “universal” antibodies and mutation‑resistant vaccines.
These findings mark a significant step toward a new generation of antiviral strategies—approaches that do not chase emerging variants, but anticipate them.
Last Updated Date : 14/05/2026



